.jpg?width=3840&quality=80&format=webp)
.jpg?width=3840&quality=80&format=webp)
Plastic
For over 40 years, we have been committed to aesthetics and reconstruction with high-quality breast implants and breast tissue expanders, science-based innovations, and the best results for patients.
MENTOR™ is purposeful in developing a comprehensive portfolio of tissue expanders and breast implants, meeting the unique and evolving needs of women and surgeons.
Featured
.jpg?width=3840&quality=90&format=webp)
Category
Breast Augmentation
Your breast augmentation patients expect only the best from you as their doctor. Trust in our proven portfolio of breast aesthetics innovations.
.jpg?width=3840&quality=90&format=webp)
Category
Breast Reconstruction
Your breast reconstruction patients envision successful outcomes. MENTOR™ offers an advanced reconstruction portfolio of high-quality breast implants and tissue expanders.1-5
.jpg?width=3840&quality=90&format=webp)
Product
MENTOR™ MemoryGel™ Enhance Breast Implants
MemoryGel Enhance Implants is the first and only family of products that supports implant-based breast reconstruction for women with larger breast sizes with volume options ranging from 930 cc to 1445 cc.6
.jpg?width=3840&quality=90&format=webp)
Resources
MENTOR™ Content Hub
Empowering your practice with digital media assets, tools, and resources.
Learning programs
Follow your unique learning journey, from foundational skills to advanced techniques, with expertly-designed programs.


SuturED
This program offers e-modules about suturing techniques and the science of tissue management.
Education library
Go deep or explore broadly through our extensive collection of high-quality learning materials.

Video
Gravity always wins - are you prepared? Dr. William P. Adams Jr. and Dr. Anand Deva
Watch Dr. William P. Adams Jr. and Dr. Deva discuss capsules and capsular contracture rates.

Video
Prepectoral breast reconstruction using MENTOR™ MemoryGel™ Enhance Implants with Risal Djohan, MD
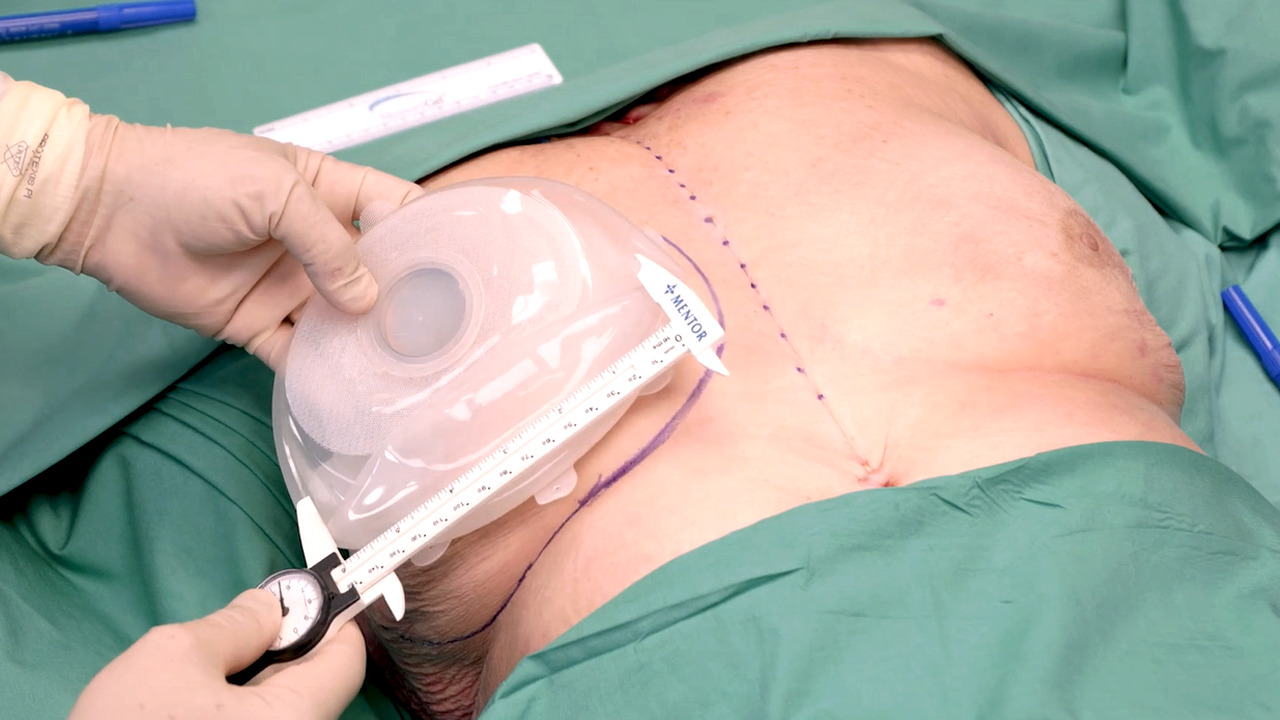
Video
Subpectoral breast reconstruction using MENTOR™ MemoryGel™ Enhance Implants with Risal Djohan, MD

Video
Look-Live Surgery™ with Dr. William P. Adams Jr. and M. Bradley Calobrace, M.D. | Primary breast augmentation with MENTOR™ MemoryGel BOOST™ Breast Implants
Elevate your expertise with J&J Institute
References
Warning
- Breast implants are not considered lifetime devices. The longer people have them, the greater the chances are that they will develop complications, some of which will require more surgery.
- Breast implants have been associated with the development of a cancer of the immune system called breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). This cancer occurs more commonly in patients with textured breast implants than smooth implants, although rates are not well defined. Some patients have died from BIA-ALCL.
- Patients receiving breast implants have reported a variety of systemic symptoms such as joint pain, muscle aches, confusion, chronic fatigue, autoimmune diseases and others. Individual patient risk for developing these symptoms has not been well established. Some patients report complete resolution of symptoms when the implants are removed without replacement.
The sale and distribution of Mentor Breast Implant Devices are restricted to users and/or user facilities that provide information to patients about the risks and benefits of the device prior to its use in the form and manner specified in approved labeling to be provided by Mentor Worldwide LLC.
Important Safety Information:
The MENTOR™ MemoryGel Enhance Breast Implants are indicated for breast reconstruction.
Breast implant surgery should not be performed in women:
- With active infection anywhere in their body
- With existing cancer or pre-cancer of their breasts who have not received adequate treatment for those conditions
- Who are currently pregnant or nursing
Safety and effectiveness have not been established in patients with autoimmune diseases (for example lupus and scleroderma), a weakened immune system, conditions that interfere with wound healing and blood clotting, or reduced blood supply to breast tissue. Patients with a diagnosis of depression, or other mental health disorders, should wait until resolution or stabilization of these conditions prior to undergoing breast implantation surgery.
Breast implants are not lifetime devices and breast implantation may not be a one-time surgery. There are risks associated with breast implant surgery. The chance of developing complications increases over time. You may need additional unplanned surgeries on your breasts because of complications or unacceptable cosmetic outcomes. Many of the changes to your breast following implantation are irreversible (cannot be undone) and breast implants may affect your ability to breastfeed, either by reducing or eliminating milk production.
The most common complications for breast reconstruction with MENTOR™ MemoryGel™ Enhance Breast Implants include any reoperation, implant removal with or without replacement, and capsular contracture. A lower risk of complication is rupture. The health consequences of a ruptured silicone gel breast implant have not been fully established. MRI screenings are recommended three years after initial implant surgery and then every two years after to detect silent rupture. Breast implants are also associated with the risk of breast implant-associated anaplastic large cell lymphoma (BIA-ALCL), an uncommon type of lymphoma. An individual's risk of developing BIA-ALCL with MENTOR™ MemoryGel™ Enhance Breast Implants is low based on the incidence of worldwide cases.
Detailed information regarding the risks and benefits associated with MENTOR™ MemoryGel™ Enhance Breast Implants is provided in MemoryGel™ Enhance Breast Implants: Important Information for Reconstruction Patients about MENTOR™ MemoryGel™ Enhance Breast Implants. These brochures are available from your surgeon or visit www.mentorwwllc.com. It is important that you read and understand these brochures when considering MENTOR™ MemoryGel™ Enhance Breast Implants.
Important Safety Information:
MENTOR CPX™4 Plus Enhance Breast Tissue Expanders are used for breast reconstruction following mastectomy, correction of an underdeveloped breast, scar revision and tissue defect procedures. The expander is intended for temporary subcutaneous or submuscular implantation and is not intended for use beyond six months. CPX™4 Plus Enhance Breast Tissue Expanders include magnetic injection domes, which contain a rare earth permanent magnet, and are NOT MRI compatible. The device could be moved by the MRI causing pain or displacement, potentially resulting in a revision surgery. Do not use the CPX™4 Plus Enhance Breast Tissue Expanders in patients where an MRI may be needed. DO NOT use the CPX™4 Plus Enhance Breast Tissue Expanders in patients that have a previously implanted device such as pacemakers, drug infusion devices, artificial sensing devices, etc. that could be affected by a magnetic field. Mentor has not tested the in vivo effects of radiation therapy with CPX™4 Plus Enhance Breast Tissue Expanders devices and cannot warrant the safety of such use. The incidence of extrusion of the expander has been shown to increase when the expander has been placed in injured areas: scarred, heavily irradiated or burned tissue, crushed bone areas, where severe surgical reduction of the area has previously been performed and where steroids are used in the surgical pocket.
Your patient needs to be informed and understand the risks and benefits of MENTOR™ CPX™4 Plus Enhance Breast Tissue Expanders and provided with an opportunity to consult with you prior to deciding on surgery.
For detailed indications, contraindications, warning and precautions associated with the use of all MENTOR™ Implantable Devices, please refer to the Instructions for Use (IFU) provided with each product, or review the Important Safety Information provided at www.mentorwwllc.com.
- Maxwell D, Estes MM, Walcott JM, et al. Safety of CPX4 Breast Tissue Expanders in Primary Reconstruction Patients. Plast Reconstr Surg Glob Open. 2021;9(3):e3425. Published 2021 Mar 26.
- Alderman A, Caplin D, Hammond DC, Keane A, Turetzky J, Kane WJ. Clinical results of mentor Memorygel Xtra breast implants from the glow clinical trial. Aesthetic Surgery Journal. 2023;43(12):NP1021-NP1032. doi:10.1093/asj/sjad272.
- Mentor US Product Catalog 2023. US_MNT_BRST_166156.
- Patient Comfort VOC March 2011.
- Bondurant, S., Ernster, V., and Herdman, R. Safety of Silicone Implants. Washington, DC: National Academy Press. 1999.
- Three-Year Interim Clinical Study Report: The Safety and Effectiveness of the Mentor® Smooth and Textured Larger Size MemoryGel® Ultra High Profile (UHP-L) Breast Implants in Subjects who are Undergoing Primary Breast Reconstruction or Revision Reconstruction ("Athena Study"). IDE Number: G150173. April 2023.
US_MNT_BRST_407206
